O–H and (CO)N–H bond weakening by coordination to Fe(ii)†
Abstract
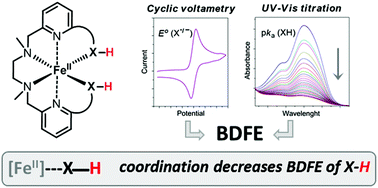
New N,N′-dimethyl-N,N′-bis(2-pyridylmethyl)-ethane-1,2-diamine derivatives bearing covalently linked OH and (CO)NH groups have been synthesized. The coordination of those pendant hydroxyl/amide groups to a Fe(II) metal center is demonstrated both in solution, even in the presence of chloride as the counterion, and in solid state, by means of X-ray diffraction crystal structures. As a result of this coordination, the experimental bond dissociation free energies (BDFE) of O–H and (CO)N–H bonds are remarkably diminished down to 76.0 and 80.5 kcal mol−1 respectively, which is also in agreement with DFT-based theoretical calculations. These BDFE values are in the range of commonly used hydrogen-atom donor reagents. The strategy presented here allows an unequivocal evaluation of the influence of metal coordination in X–H bond weakening in organic solvents which could be easily extended to other metal centers.


 Please wait while we load your content...
Please wait while we load your content...