Modulation of the reactivity of nitrogen mustards by metal complexation: approaches to modify their therapeutic properties
Abstract
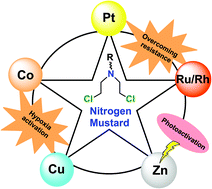
The alkylating agents bearing the –N(CH2CH2Cl)2 moiety, commonly known as ‘the nitrogen mustards,’ are among the first chemotherapeutic agents against cancer. They form covalent alkyl linkages due to reaction with nucleophilic entities viz. N7 of guanine in DNA. The reactivity of nitrogen mustards may be controlled in various ways, which include metabolic activation, reductive or hypoxic activation and metal complexation. This review discusses how the metal complexation of nitrogen mustards affects their reactivity and mechanistic pathways. The discussion encompasses those transition metal complexes for which the structure has been well characterized and cytotoxicity studies have been performed. This review discusses how the binding of the metal centre along with its oxidation state helps to control the reactivity of the nitrogen mustards. The discussion emphasizes the effect of the reduction potential of the coordinated metal center on the reactivity of the respective mustard under specific conditions, the dependence of efficiency and specificity on the stability of the reduced species and the importance of steric hindrance around the metal center. The insight into the mechanism of action is expected to provide a better understanding to overcome the existing lacunae and design better molecules of this class which have excellent potential, given the ever growing need for therapeutics against cancer.
- This article is part of the themed collection: 2019 Frontier and Perspective articles


 Please wait while we load your content...
Please wait while we load your content...