The influence of ZnII coordination sphere and chemical structure over the reactivity of metallo-β-lactamase model compounds†
Abstract
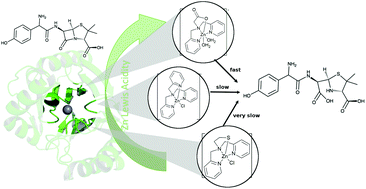
A systematic study of the influence of the first coordination sphere over the reactivity and structure of metallo-β-lactamase (MβL) monozinc model complexes is reported. Three ZnII complexes with tripodal ligands forming the series [Zn(N-NNN)], [Zn(N-NNS)], and [Zn(N-NNO)] where N-NNX represents the tripodal donor atoms were investigated regarding their ability to mimic MβL. The tripodal series was inspired by MβL active sites in the respective subclasses, representing the (His, His, His) Zn1 site present in B1 and B3 subclasses, (His, His, Asp) present in the B3 subclass site and the thiolate present in B1 and B2 sites. The results were supported by electronic structure calculations. XAS analysis demonstrated that the ZnII electronic deficiency significantly changes in the order [Zn(N-NNS)] < [Zn(N-NNN)] < [Zn(N-NNO)]. This effect directly affects the reactivity over nitrocefin and amoxicillin, observed by the hydrolysis kinetics, which follows the same trend. NMR spectroscopy revealed the coordination of the carboxylic group in the substrate to the metal changes accordingly, affecting the hydrolysis kinetics. Our results also demonstrated that not only the Lewis acidity is changed by the ligand system but also the softness of the metal. [Zn(N-NNS)] is softened by the thiolate, promoting the ligand substitution reaction with solvents and favoring a secondary interaction with substrates, not observed for [Zn(N-NNO)]. XRD of the models reveals their similar geometric aspects in comparison to the crystal structure of GOB MβL. The present work demonstrates that the ZnII electronic details must be considered in the design of new MβL models that will further aid in the design of clinically useful inhibitors.


 Please wait while we load your content...
Please wait while we load your content...