Room temperature CO oxidation catalysed by supported Pt nanoparticles revealed by solid-state NMR and DNP spectroscopy†
Abstract
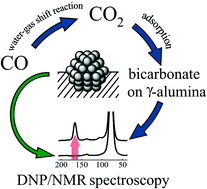
A series of 1 and 2 nm sized platinum nanoparticles (Pt-NPs) deposited on different support materials, namely, γ-alumina (γ-Al2O3), titanium dioxide (TiO2), silicon dioxide (SiO2) and fumed silica are investigated by solid-state NMR and dynamic nuclear polarization enhanced NMR spectroscopy (DNP). DNP signal enhancement factors up to 170 enable gaining deeper insight into the surface chemistry of Pt-NPs. Carbon monoxide is used as a probe molecule to analyze the adsorption process and the surface chemistry on the supported Pt-NPs. The studied systems show significant catalytic activity in carbon monoxide oxidation on their surface at room temperature. The underlying catalytic mechanism is the water–gas shift reaction. In the case of alumina as the support the produced CO2 reacts with the surface to form carbonate, which is revealed by solid-state NMR. A similar carbonate formation is also observed when physical mixtures of neat alumina with silica, fumed silica and titania supported Pt-NPs are studied.


 Please wait while we load your content...
Please wait while we load your content...