The electrooxidation of formic acid catalyzed by Pd–Ga nanoalloys†
Abstract
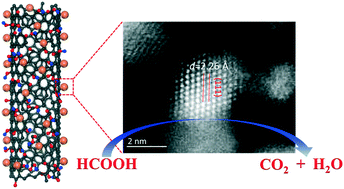
In the present work, PdxGay alloys on oxygen-species-functionalized carbon nanotubes (PdxGay/OCNTs) are prepared as catalysts for the electrooxidation of formic acid by a facile method. Among all the catalysts, the catalyst with a Pd : Ga atomic ratio of 3.9 : 1 (Pd3.9Ga1) exhibits mass activity of 1321 A g−1 and improved stability. The functional groups on the modified CNT surfaces provide anchoring sites for Pd3.9Ga1 nanoalloys of 2 nm average size. Surface analysis reveals the appearance of a Pd 4d band center shift, which may weaken the adsorption of the formate intermediate. Pd3.9Ga1/OCNT with a modified electronic structure shows enhanced catalytic activity and stability towards the HCOOH oxidation reaction.


 Please wait while we load your content...
Please wait while we load your content...