Activity enhancement of Pt/MnOx catalyst by novel β-MnO2 for low-temperature CO oxidation: study of the CO–O2 competitive adsorption and active oxygen species†
Abstract
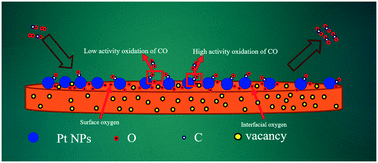
Low-temperature oxidation of carbon monoxide (CO) is one of the most studied reactions because of its scientific and industrial applications. In this study, we discovered that the low-temperature CO oxidation activity over Pt/MnOx catalyst could be enhanced by a novel β-MnO2 support material. Nearly complete CO conversion was obtained over the Pt/MnOx catalyst below 100 °C under our catalysis conditions. TOF (turnover frequency) of Pt/MnOx was five and nine times that of Pt/MnOx-COM (COM, commercial) and Pt/Al2O3 at 150 °C, respectively. The combined results of X-ray diffraction (XRD) Rietveld refinement of fresh and spent catalysts, CO-temperature-programmed desorption (CO-TPD) and diffuse reflectance infrared Fourier transform (DRIFT) spectroscopy allowed us to conclude that the O2 molecules exhibited strong adsorption and activation ability on the surface of Pt/MnOx compared to that on the surface of Pt/MnOx-COM. From the H2-TGA (weight loss) and DTGA results, we confirmed that strong metal–support interactions exist between Pt and MnOxvia formation of Mn–O–Pt bonds for both catalysts. Moreover, by combining the results of Rietveld refinement of XRD, Raman, CO-TPD and O2-TPD (inlet CO and inlet CO/O2 mixture gases as adsorbates, respectively) analyses, we inferred that the interfacial lattice oxygen in Pt/MnOx might be the active oxygen species for low-temperature CO oxidation.


 Please wait while we load your content...
Please wait while we load your content...