Comparison of TiO2 and g-C3N4 2D/2D nanocomposites from three synthesis protocols for visible-light induced hydrogen evolution†
Abstract
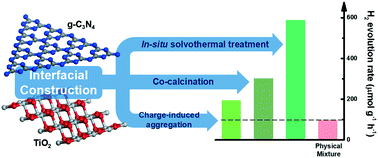
Knowledge of the interfacial structure of nanocomposite materials is a prerequisite for rational design of nanostructured photocatalysts. Herein, TiO2 and g-C3N4 2D/2D nanocomposites were fabricated from three distinct synthetic protocols (i.e., co-calcination, solvothermal treatment and charge-induced aggregation), showing different degrees of enhancement (1.4–6.1 fold) in the visible-light induced photocatalytic hydrogen evolution reaction compared to the simple physical mixture. We propose that the interfacial Ti–O–N covalent bonding promotes the charge carrier transfer and separation more effectively than the electrostatic interaction, thus accelerating the photocatalytic H2 production. Meanwhile, the exposed surface area of TiO2 in the composite needs to be enlarged for deposition of the co-catalyst. This research sheds light on the rational design of hybrid nanocomposites based on earth-abundant elements for photocatalysis.


 Please wait while we load your content...
Please wait while we load your content...