Enhancement of metallo-supramolecular dissociation kinetics in telechelic terpyridine-capped poly(ethylene glycol) assemblies in the semi-dilute regime†
Abstract
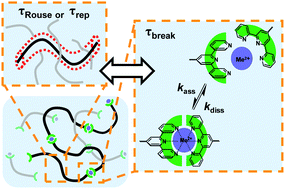
The dynamics of supramolecular polymer assemblies is governed by that of their polymeric building blocks and that of the transient bonds between them. Entrapment of such bonds by topological crowding often causes renormalization of the bond lifetimes towards prolonging. In the present study, by contrast, we show that this effect can also be inverse in the case of telechelic metallo-supramolecular polymers in semi-dilute solution. We focus on linear poly(ethylene glycols) capped by terpyridine binding motifs at both ends that can form metal–ligand coordinative bonds with various transition metal ions, thereby creating transient metallo-supramolecular assemblies of varying length and binding strength. Oscillatory shear rheology measurements along with theoretical modelling of the mechanical spectra of these samples reveals a pronounced enhancement of the complex dissociation kinetics that is dependent on the length of the polymeric chain segment, with longer segments yielding faster dissociation times up to six orders of magnitude shorter than described for the free complexes. This finding indicates that the dynamic activity of the polymer chain itself causes complex destabilization.


 Please wait while we load your content...
Please wait while we load your content...