Porosity controls the catalytic activity of platinum nanoparticles†
Abstract
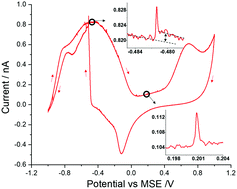
Dendritic/mesoporous nanoparticle structures arise naturally and result from aggregation based growth mechanisms. The resulting porous particles exhibit high total surface areas (internal and external) but determining the magnitude of the interface remains challenging. Furthermore, assessing the chemical accessibility of the catalytic interface presents an additional difficulty. Taking three structurally related but different sized platinum nanoparticle samples (30–70 nm), we demonstrate how the catalytic rate of two archetypal surface limited reactions scale not with the square of the particle radius but with a power law of 2.6–2.9. This power law directly reflects the mesoporosity of the nanoparticles; the internal surface of the nanoparticles is both chemically accessible and contributes to the catalytic activity. For the 70 nm particles, up to 60% of the catalytic surface is contained in the internal structure of the particle.


 Please wait while we load your content...
Please wait while we load your content...