Ab initio study of a 2D h-BAs monolayer: a promising anode material for alkali-metal ion batteries†
Abstract
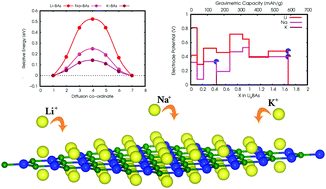
The selection of a suitable two dimensional anode material is one of the key steps in the development of alkali metal ion batteries to achieve superior performance with an ultrahigh rate of charging/discharging capability. Here, we have used state of the art density functional theory (DFT) to explore the feasibility of two dimensional (2D) honeycomb boron arsenide (h-BAs) as a potential anode for alkali-metal (Li/Na/K)-ion batteries. The structural and dynamic stability has been confirmed from the formation energy and the non-negative phonon frequency. The h-BAs monolayer exhibits negative adsorption-energy values of −0.422, −0.321 and −0.814 eV, for the Li, Na, and K-ions, respectively. Subsequently, during the charging process the adsorption-energy increases considerably without an energy-barrier when any of the A-atoms achieve a crucial distance (∼8 Å). In addition, it has been observed that insertion of the mono alkali metal atom into the h-BAs surface results in the semi-conducting nature of the monolayer being transformed into a metallic-state. The low energy barriers for Li (0.522 eV), Na (0.248), and K (0.204 eV) active ion migration imply high diffusion over the h-BAs surface, hence suggesting it has a high charge/discharge capability. Moreover, we have obtained low average operating voltages of 0.49 V (Li), 0.35 V (Na) and 0.26 V (K) and high theoretical capacities of 522.08 mA h g−1 (for Li and Na) and 209.46 mA h g−1 (for K) in this study. The aforementioned findings indicate that a h-BAs monolayer could be a promising anode material in the search for low cost and high performance alkali metal ion batteries.


 Please wait while we load your content...
Please wait while we load your content...