Exciton dissociation in an NIR-active triohybrid nanocrystal leading to efficient generation of reactive oxygen species
Abstract
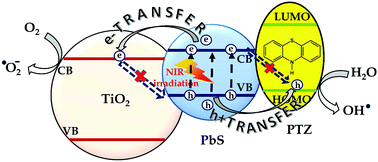
Lead sulfide (PbS) colloidal quantum dots (QDs) are emerging materials for fundamental studies because of their potential application in near infrared (NIR) light harvesting technologies. However, inefficient electron separation, facile charge recombination and defect state trapping of photoexcited carriers are reported as limitations of the PbS QDs to achieve efficient energy conversion. In the present study, we have synthesized a triohybrid by assembling a semiconductor titanium dioxide (TiO2), an organic oxidizing molecule phenothiazine (PTZ) and PbS QDs. The triohybrid along with PbS_TiO2 and PbS_PTZ hybrids has been characterized and the attachment of different components is verified by spectroscopic and microscopic techniques. The interfacial dynamics of the photoexcited carriers in the PbS_TiO2 and PbS_PTZ hybrids have been investigated separately using steady state and time resolved photoluminescence (TRPL) measurements. The photoinduced electron transfer (PET) from the PbS QD to the conduction band (CB) of TiO2 and photoinduced hole transfer (PHT) from the valence band (VB) of the QD to the highest occupied molecular orbital (HOMO) of PTZ have been observed and correlated mechanistically to the energy level alignments obtained from cyclic voltammetric (CV) analysis. The PTZ molecule is also found to act as a surface defect passivator of the PbS QD. Finally, simultaneous exciton dissociation and reduced back recombination phenomena have been correlated with a higher reactive oxygen species (ROS) generation activity of the triohybrid than the other two, under IR light irradiation. Thus, a detailed investigation of carrier dynamics and the mechanism of higher NIR light activity for a novel nanohybrid is explored and analyzed which could be beneficial for NIR catalysis or antibacterial activities.


 Please wait while we load your content...
Please wait while we load your content...