UO22+ structure in solvent extraction phases resolved at molecular and supramolecular scales: a combined molecular dynamics, EXAFS and SWAXS approach†
Abstract
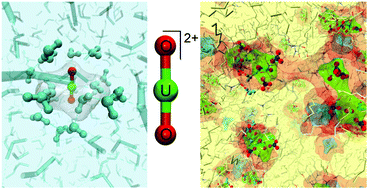
A new polarizable force field for describing the solvation of the uranyl (UO22+) cation in solvent extraction phases has been developed for molecular dynamics simulations. The validity of the polarizable force field has been established by comparison with EXAFS and SWAXS experiments. This new force field allows for describing both the UO22+ hydration and solvation properties in good agreement with the experiments. In aqueous phases we demonstrated that the UO22+ force field has been improved from the previous one we developed. Indeed, the UO22+ structural and dynamics properties, i.e., the dynamics of the water molecules in the vicinity of the uranyl cation, calculated from molecular dynamics are in better agreement with the EXAFS experiments. Furthermore, the transferability of the UO22+ force field proposed here has been validated on typical solvent extraction phases containing uranyl nitrate salts with extractant molecules, namely DMDOHEMA molecules, in n-heptane. The good agreements observed between the theoretical (MD simulations) and experimental UO22+ structures at the molecular (EXAFS) and supramolecular (SWAXS) scales prove the accuracy of the UO22+ force field developed and proposed in the present paper.


 Please wait while we load your content...
Please wait while we load your content...