Interfacial premelting of ice in nano composite materials†
Abstract
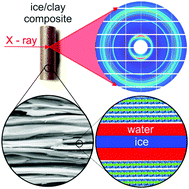
The interfacial premelting in ice/clay nano composites was studied by high energy X-ray diffraction. Below the melting point of bulk water, the formation of liquid water was observed for the ice/vermiculite and ice/kaolin systems. The liquid fraction is gradually increasing with temperature. For both minerals, similar effective premelting layer thicknesses of 2–3 nm are reached 3 K below the bulk melting point. For the quantitative description of the molten water fraction in wet clay minerals we developed a continuum model for short range interactions and arbitrary pore size distributions. This model quantitatively describes the experimental data over the entire temperature range. Model parameters were obtained by fitting using a maximum entropy (MaxEnt) approach. Pronounced differences in the deviation from Antonow's rule relating interfacial free energy between ice, water, and clay are observed for the charged vermiculite and uncharged kaolin minerals. The resultant parameters are discussed in terms of their ice nucleation efficiency. Using well defined and characterized ice/clay nano composite samples, this work bridges the gap between studies on single crystalline ice/solid model interfaces and naturally occurring soils and permafrost.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...