Investigation of solid–liquid phase diagrams of the sulfamethazine–salicylic acid co-crystal†
Abstract
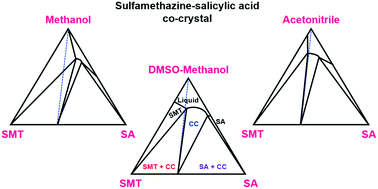
The influence of temperature and solvent on the solid–liquid phase diagram of the 1 : 1 sulfamethazine–salicylic acid co-crystal has been investigated. Ternary phase diagrams of this co-crystal system have been constructed in three solvents: methanol, acetonitrile and a 7 : 3 (v/v) dimethylsulfoxide–methanol mixture, at three temperatures. The system exhibits congruent dissolution in acetonitrile and the co-crystal solubility has been determined by a gravimetric technique. The Gibbs energy of co-crystal formation from the respective solid components has been estimated from solubility data, together with the corresponding enthalpic and entropic component terms. The Gibbs energy of formation ranges from −5.7 to −7.7 kJ mol−1, with the stability increasing with temperature. In methanol and the DMSO–methanol mixture, the co-crystal dissolves incongruently. It is shown that the solubility ratio of the pure components cannot be used to predict with confidence whether the co-crystal will dissolve congruently or incongruently. The size of the region where the co-crystal is the only stable solid phase is inversely related to the pure component solubility ratio of salicylic acid and sulfamethazine.


 Please wait while we load your content...
Please wait while we load your content...