The interplay among molecular structures, crystal symmetries and lattice energy landscapes revealed using unsupervised machine learning: a closer look at pyrrole azaphenacenes†
Abstract
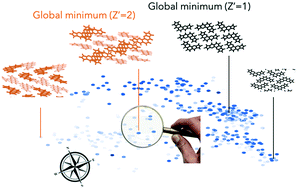
The ability to perform large-scale crystal structure predictions (CSPs) has significantly advanced the synthesis of functional molecular solids by design. In our recent work [J. Yang, S. De, J. E. Campbell, S. Li, M. Ceriotti and G. M. Day, Chem. Mater., 2018, 30, 4361], we demonstrated our latest developments in organic CSPs by screening a set of 28 pyrrole azaphenacene isomers which led to one new molecule with higher thermodynamic stability and carrier mobilities in its crystalline form, compared to the one reported experimentally. Hereby, using the lattice energy landscapes of pyrrole azaphenacenes as examples, we applied machine-learning techniques to statistically reveal, in more detail, how molecular symmetry and Z′ values translate to the crystal packing landscapes, which in turn affect the coverage of landscapes through quasi-random crystal structure samplings. A recurring theme in crystal engineering is to identify the probabilities of targeting isostructures to a specific reference crystal upon chemical functionalisations. For this, we propose here a global similarity index in conjunction with an energy–density–isostructurality (EDI) map to analyse the lattice energy landscapes of halogen substituted pyrrole azaphenacenes. A continuous effort in the field is to accelerate CSPs for sampling a much wider chemical space for high-throughput material screenings, and we propose a potential solution to this challenge drawn upon this study. Our work will hopefully stimulate the crystal engineering community in adapting a more statistically-oriented approach in understanding the crystal packing of organic molecules in the age of digitisation.
- This article is part of the themed collection: Editor’s Collection: Computer aided solid form design


 Please wait while we load your content...
Please wait while we load your content...