Insight into the optical, color, photoluminescence properties, and photocatalytic activity of the N–O and C–O functional groups decorating spinel type magnesium aluminate†
Abstract
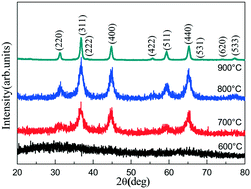
MgAl2O4 with a spinel structure was successfully synthesized using a gamma-ray irradiation assisted polyacrylamide gel method. The samples were synthesized at different calcining temperatures to study the effects on the phase purity, optical, color and fluorescence properties and photocatalytic activity of the as-prepared samples. X-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FTIR) analysis indicates that the calcining temperature has an obvious influence on the crystallinity and the formation of the MgAl2O4 spinel phase. Transmission electron microscopy (TEM) observation shows that the primary crystal of the MgAl2O4 nanoparticles is only 30 nm and exhibits a higher uniformity than those of MgAl2O4 nanoparticles fabricated using the conventional polyacrylamide gel method. Optical properties show that the optical energy gap (Eg) of the MgAl2O4 nanoparticles increases with the increasing calcining temperature, mainly due to the interaction between the N–O and C–O functional groups and the calcining temperature. An abnormal result for the color parameter of the MgAl2O4 xerogel powders calcined at 900 °C indicates the enhancement of the crystallinity and the phase purity of the MgAl2O4 nanoparticles. The fluorescence spectra indicates that the emission intensity of the emission peak at 395 nm decreases with the increasing calcining temperature. The photocatalytic activity of the MgAl2O4 nanoparticles prepared at different calcining temperature were then tested for photocatalytic degradation of the methylene blue (MB) dye. The result indicates that the MgAl2O4 xerogel powders calcined at 800 °C exhibit the highest photocatalytic activity, and that this is relevant to the light absorption ability, band edge position, separation and utilization efficiency of the photogenerated carriers of the as-prepared sample. The fluorescence and photocatalytic mechanisms show that a small number of N–O and C–O functional groups in the presence of the MgAl2O4 nanoparticles can help to improve the fluorescence properties and photocatalytic activity of the MgAl2O4 nanoparticles.


 Please wait while we load your content...
Please wait while we load your content...