Modulation of catalyst enantioselectivity through reversible assembly of supramolecular helices†
Abstract
A multi-configurable catalyst, for which the degree of enantioinduction in successive reactions is varied between 6% ee and 52% ee, is achieved by supporting copper centres at the periphery of supramolecular helices. Precise characterization of the co-assemblies corroborates the relation between helix length and catalyst selectivity.
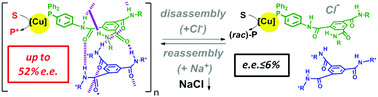
- This article is part of the themed collection: Switchable Catalysis


 Please wait while we load your content...
Please wait while we load your content...