Two RP-HPLC assay methods with different chromatographic approaches for the simultaneous estimation of bambuterol and its main degradation product, terbutaline†
Abstract
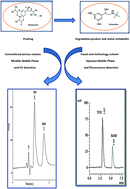
The present study describes two stability-indicating HPLC methods with two different chromatographic approaches for the simultaneous estimation of bambuterol and its main degradation product, terbutaline in bulk and pharmaceuticals. Method I was carried out using a conventional particle-packed cyano column (150 mm × 4.6 mm, 5 μm particle size) and a micellar eluent composed of 0.1 M SDS, 15% n-propanol, and 0.3% triethylamine (pH 3.5), coupled with UV detection at 220 nm. Method II was based on the fused core shell technology, and the separation was carried out using a Kinetex C18 column (150 mm × 4.6 mm, 5 μm particle size) and an aqueous mobile phase composed of methanol and a 0.1 M phosphate buffer (45 : 55 v/v; pH 4), using fluorescence detection at λex = 275 nm and λem = 313 nm. The first method was rectilinear over the concentration range of 10–200 μg mL−1 for bambuterol and 5–100 μg mL−1 for terbutaline while the second method showed a good linearity in the range of 1–5 μg mL−1 for bambuterol and 0.1–1 μg mL−1 for terbutaline. The developed methods enabled the quantification of bambuterol and terbutaline in their dosage forms, including tablets and syrups and showed good results compared to the official methods. The practical and analytical comparisons between the two proposed methods is discussed.


 Please wait while we load your content...
Please wait while we load your content...