Dual role of BSA for synthesis of MnO2 nanoparticles and their mediated fluorescent turn-on probe for glutathione determination and cancer cell recognition†
Abstract
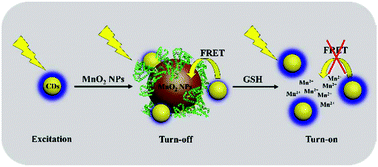
A MnO2 nanoparticle (MnO2 NP)-mediated fluorescent turn-on probe for sensitively and selectively detecting glutathione (GSH) and recognizing cancer cells was established in this work. MnO2 NPs were synthesized simply and quickly through an in situ redox reaction by mixing bovine serum albumin (BSA) and KMnO4, in which BSA served the dual roles of template and reductant. It was found that the MnO2 NPs served as an effective energy acceptor and quenched the fluorescence intensity of carbon dots (CDs), owing to the fluorescence resonance energy transfer (FRET) process. Further, the addition of GSH triggered the decomposition of MnO2, breaking the FRET between MnO2 NPs and CDs and thus restoring the fluorescence intensity of CDs. Based on this mechanism, quantitative determination of GSH was performed. Under optimal conditions, a satisfactory linear range of 0.05–90 μM and limit of detection of 39 nM were obtained, and GSH content in human serum samples was detected. Moreover, taking advantage of the higher levels of GSH in cancer cells than in normal cells, the MnO2 NP-CD probe was applied to distinguish SMMC-7721 cancer cells from L02 normal cells. The FRET was interrupted by GSH in cancer cells, and strong fluorescence was observed. This work provides a facile approach for synthesizing MnO2 NPs, and this rapid, low-cost method with no need for reductants makes synthesis green and convenient. The MnO2 NP-mediated fluorescent turn-on response to GSH could improve the MnO2 nanomaterial-based biochemical analysis applications.
- This article is part of the themed collections: In celebration of Chinese New Year 2020 and Bioanalytical Sensors


 Please wait while we load your content...
Please wait while we load your content...