Oxidative stress-mediated apoptotic cell death induced by camphor in sod1-deficient Schizosaccharomyces pombe
Abstract
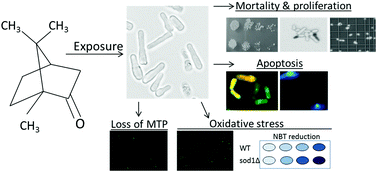
Camphor is one of the monoterpenes widely used in cosmetics, pharmaceutics and the food industry. In this study, we aimed to assess the oxidative, cytotoxic and apoptotic effects of camphor on the fission yeast (Schizosaccharomyces pombe), which is a promising unicellular model organism in mechanistic toxicology and cell biology. Since Sod1 is the main radical scavenger in the cell, we used sod1 mutants to understand whether camphor-induced ROS accumulation caused higher cytotoxicity and apoptosis. Camphor exposure (0–2000 mg L−1) caused significant cytotoxicity in yeast, particularly in sod1Δ cells. DCFDA (2,7-dichlorodihydrofluorescein diacetate) fluorescence and NBT (p-nitro-blue tetrazolium chloride) reduction increased (at least 2.5–3-fold in sod1Δ cells) in correlation with camphor concentrations (800–1200 mg L−1), showing higher ROS levels and oxidative stress. Moreover, cells, stained with acridine orange/ethidium bromide, showed an apoptotic morphology with nuclear fragmentation and condensation. DAPI (4′,6-diamidino-2-phenylindole) staining was used to validate the apoptotic nuclear morphology. Dramatically increased mitochondrial impairment, which was higher in sod1Δ cells than in wild type cells, was shown by rhodamine 123 staining. In conclusion, camphor-induced excessive ROS production, which could not be prevented significantly in sod1 mutants, caused a dramatic increase in mortality rates due to intrinsic apoptosis revealed by mitochondrial impairment and apoptotic nuclear morphology. The potential effects of camphor on apoptotic cell death and the underlying mechanisms were clarified in the unicellular eukaryotic model, S. pombe.


 Please wait while we load your content...
Please wait while we load your content...