A simple model to solve a complex drug toxicity problem†
Abstract
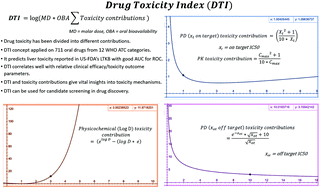
Linear drug toxicity models like therapeutic index (TI), physicochemical rules (rule of five, 3/75), ligand efficiency indices (LEI), ideal pharmacokinetic (PK) and pharmacodynamic (PD) profiles are widely used in drug discovery and development. In spite of this, predicting drug toxicity at various stages remains challenging and the overall productivity (<20%) and ultimate benefit to the patients remain low. A simple drug toxicity model, “Drug Toxicity Index” (DTI), is developed here using 711 oral drugs. DTI redefines drug toxicity as scaled biphasic and exponential functions of PD, PK and physicochemical parameters. PD, PK and physicochemical toxicity contributions were estimated from the on and off target IC50, maximum unbound plasma drug concentration (free Cmax), and log D values, respectively. These contributions are then scaled by molar dose and oral bioavailability and the logarithm of the sum of scaled contributions is DTI. Drugs with DTI above the WHO ATC drug category specific average values consistently have toxic profiles, while drugs with DTI below this average are relatively safe. DTI performs better than standard rules for lead optimization, LEI and exposure based TIs in identifying safe and toxic drugs. DTI classifies 392 drugs reported in the US-FDA's Liver Toxicity Knowledge Base (LTKB) with an AUC for ROC curves of 0.91–0.64 for different WHO ATC categories. DTI has been used to predict network meta-analysis results on relative toxicity within/across eight different therapeutic areas. It is useful in understanding PD, PK and physicochemical toxicity contributions and identifying potentially toxic drugs and the toxicity of recently approved drugs. Decision trees are proposed for applying the DTI concept in preclinical drug discovery and clinical trial settings. DTI can potentially reduce failure in drug discovery and might be useful in therapeutic drug monitoring and in xenobiotic and environmental toxicity studies.
- This article is part of the themed collection: Toxicology Research Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...