Diverse sensor responses from two functionalized tris(phthalocyaninato)europium ambipolar semiconductors towards three oxidative and reductive gases†
Abstract
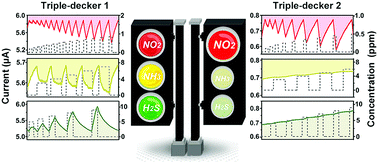
Ambipolar tris(phthalocyaninato) rare earth semiconductors, Eu2[Pc(OC8H17)8]2[Pc(OCH2CF3)8] (1) and Eu2{Pc[(OC2H4)3OCH3]8}2[Pc(OCH2CF3)8] (2), are synthesized and fabricated into well-organized films via a Langmuir-Shäfer (LS) method. Driven by intermolecular π–π stacking, as well as hydrophilic–hydrophobic interactions, 1 and 2 aggregated in different modes, H for 1 and J for 2, in LS films. Consequently, higher conductivity and carrier mobilities for electrons and holes in the LS films of 1 are achieved compared to those of 2, with gate-induced sheet electron and hole densities (ne and nh) of 2.48 × 1012 and 8.31 × 109 cm−2 for 1, and 1.77 × 1012 and 8.01 × 1010 cm−2 for 2, respectively. Comparative studies on the gas sensing behavior of the LS films of 1 and 2 revealed that, towards oxidative NO2, both 1 and 2 show n-type sensing behavior, with a higher sensitivity of 2 (54.6% ppm−1) than 1 (12.5% ppm−1) in the 0.10–0.85 ppm range, which can be attributed to the lower ne of 2. Unexpectedly, towards reductive NH3 (3.0–7.0 ppm) and H2S (5.0–9.0 ppm), only ambipolar 1 exhibits current responses, determined by the lower nh of 1. More importantly, the current of 1 responds reversely towards NH3 and H2S (decreases vs. increases) even though they are both reductive gases. This is because weakly reductive NH3 reduces nh while strongly reductive H2S increases ne. By combining the distinct response patterns of ambipolar 1 and 2, these three toxic gases can be distinguished unambiguously from each other, showing the potential for these two molecules in efficient chemiresistor sensor arrays.


 Please wait while we load your content...
Please wait while we load your content...