Electric field–temperature phase diagrams for (Bi1/2Na1/2)TiO3–BaTiO3–(K1/2Na1/2)NbO3 relaxor ceramics
Abstract
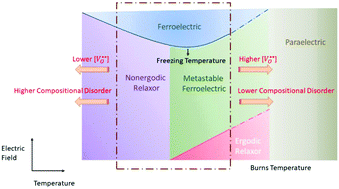
The electric field–temperature (E–T) phase diagrams for three model compositions in the lead-free (Bi1/2Na1/2)TiO3–BaTiO3–(K1/2Na1/2)NbO3 (BNT–BT–KNN) relaxor system have been constructed, to serve as a template for other similar systems. The compositions chosen are known to exhibit nonergodic (BNT–6BT), ergodic (BNT–6BT–3KNN) and ferroelectric (BNT–12BT) behaviors at room temperature. Through temperature-dependent permittivity and piezoelectric measurements, the transition pathways across relaxor–ferroelectric states were determined as functions of temperature and electric field. The relaxor compositions were found to have a typical U-shaped E–T phase diagram and exhibited typical transitions to the ferroelectric phase upon applying electric fields above threshold values. BNT–6BT is suggested to have a freezing temperature (Tf) of approximately 120 °C and exhibited a “mixed” transition behavior between 90 °C and 120 °C, characterized by the presence of both partially pinched polarization hysteresis loops, characteristic of ergodic relaxors, and substantial negative strains, indicating a nonergodic behavior. Similar behavior was observed for BNT–6BT–3KNN, which had a rather broad temperature range (21 °C to −40 °C) where it exhibited the “mixed” transition behavior. The field required for the relaxor to ferroelectric transition was 20 kV cm−1 and 30 kV cm−1 for BNT–6BT and BNT–6BT–3KNN at Tf, respectively. BNT–12BT was ferroelectric at room temperature, however it transformed spontaneously to an ergodic relaxor at approximately 200 °C upon heating. Additionally, to study the influence of point defects on the E–T phase diagrams, nonstoichiometry was intentionally introduced to alter the oxygen vacancy concentration in the BNT–6BT composition. It was found that lowering the oxygen vacancy concentration lowers Tf, while increasing their concentration has the opposite effect. This provides an avenue to tune crucial materials’ parameters such as Tf and critical field values and modify their structural transition pathways to suit application requirements.


 Please wait while we load your content...
Please wait while we load your content...