Preparation, electronic structure of gadolinium oxyhydride and low-energy 5d excitation band for green luminescence of doped Tb3+ ions†
Abstract
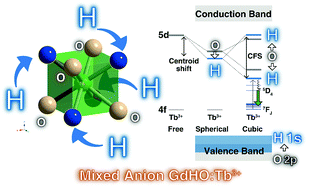
Oxyhydride compounds of GdHO and GdHO:Tb3+ were successfully prepared by solid state reaction of GdH3, Gd2O3 and Gd2O3:Tb3+ under a H2 atmosphere. XRD analysis suggests that GdHO has a cubic fluorite structure (space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, lattice parameter a = 5.38450(4) Å for GdHO and 5.38523(6) Å for GdHO:Tb), which is different from the tetragonal fluorite-like superstructure for previously reported LnHO (Ln = La, Ce, Pr and Nd) (space group P4/nmm) materials. The prepared GdHO:Tb3+ shows photoluminescence bands in the range between 470 and 650 nm, which are attributed to Tb3+:5D4 → 7FJ transitions. In the photoluminescence excitation spectrum, a broad 4f–5d band of Tb3+ was clearly observed at around 310 nm, which is one of the lowest 5d energy positions compared with other Tb3+-doped compounds. This shift may be caused by partially coordinated hydride ions with strong covalency around Tb3+, which cause a large nephelauxetic effect and crystal field splitting. The lifetime of Tb3+:5D4 is almost unchanged from 83 to 500 K and decreases above 500 K. The thermal luminescence quenching of the 5D4 → 7FJ transition is caused by a thermally activated crossover to the ground state through the 5d energy state. The band gap energy of GdHO (5.39 eV) was smaller than that of cubic and monoclinic Gd2O3 (5.93 and 6.10 eV, respectively). Based on the constructed vacuum referred binding energy diagram and DFT calculation, the top of the valence band (VB) energy of GdHO is much higher than that of cubic and monoclinic Gd2O3 due to the higher energy level of the hydride 1s orbital than that of the oxide 2p orbital.
m, lattice parameter a = 5.38450(4) Å for GdHO and 5.38523(6) Å for GdHO:Tb), which is different from the tetragonal fluorite-like superstructure for previously reported LnHO (Ln = La, Ce, Pr and Nd) (space group P4/nmm) materials. The prepared GdHO:Tb3+ shows photoluminescence bands in the range between 470 and 650 nm, which are attributed to Tb3+:5D4 → 7FJ transitions. In the photoluminescence excitation spectrum, a broad 4f–5d band of Tb3+ was clearly observed at around 310 nm, which is one of the lowest 5d energy positions compared with other Tb3+-doped compounds. This shift may be caused by partially coordinated hydride ions with strong covalency around Tb3+, which cause a large nephelauxetic effect and crystal field splitting. The lifetime of Tb3+:5D4 is almost unchanged from 83 to 500 K and decreases above 500 K. The thermal luminescence quenching of the 5D4 → 7FJ transition is caused by a thermally activated crossover to the ground state through the 5d energy state. The band gap energy of GdHO (5.39 eV) was smaller than that of cubic and monoclinic Gd2O3 (5.93 and 6.10 eV, respectively). Based on the constructed vacuum referred binding energy diagram and DFT calculation, the top of the valence band (VB) energy of GdHO is much higher than that of cubic and monoclinic Gd2O3 due to the higher energy level of the hydride 1s orbital than that of the oxide 2p orbital.


 Please wait while we load your content...
Please wait while we load your content...