Intracellular delivery of a native functional protein using cell-penetrating peptide functionalized cubic MSNs with ultra-large mesopores†
Abstract
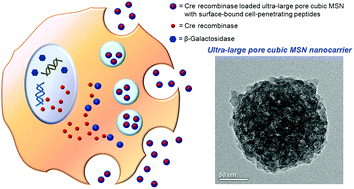
The intracellular delivery of functional proteins in their native forms into cells is a theme of paramount importance in research owing to their diverse biological applications. Porous inorganic nanoparticles are emerging as efficient nanocarriers for the delivery of small molecules and drugs. To expand the range of cargos from small molecules to large native functional proteins, cubic mesoporous silica nanoparticles (cMSNs) with a Pm3n pore symmetry with an average particle dimension of 180 nm were prepared. The as-prepared cMSNs were subsequently etched with a methanolic solution of Ca(NO3)2 to expand their mesopores and simultaneously remove the template. The original mesopores with a pore dimension of 2.41 nm partially collapsed and combined into ultra-large mesopores with an average pore diameter of 13.89 nm without perturbing the original cubic symmetry of the remaining mesopores. The maximum pore dimension was around 60 nm. Various techniques including powder X-ray diffraction, transmission electron microscopy, and electron tomography identified the unique three-dimensional structure of pore-enlarged cMSNs (Ca-cMSNs). Moreover, their surfaces were functionalized with a guanidinium-rich cell-penetrating R8-azido-peptide (p-azidophenylalanine-GSGSGGRRRRRRRR) through the click reaction. The intracellular delivery of functional proteins such as Cre recombinase into human TE671(LoxP-LacZ) cells was realized by using R8-Ca-cMSNs as native protein delivery synthetic nanocarriers. The delivery efficiency when using the R8-Ca-cMSNs significantly enhanced compared to that when using Ca-cMSNs without surface-bound cell-penetrating peptides.


 Please wait while we load your content...
Please wait while we load your content...