Polypeptide nano-Se targeting inflammation and theranostic rheumatoid arthritis by anti-angiogenic and NO activating AMPKα signaling pathway†
Abstract
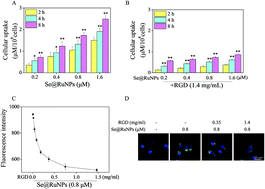
Rheumatoid arthritis (RA) is a chronic autoimmune disease and there is a lack of effective treatments. Nitric oxide (NO) plays an important role in inflammatory diseases, but the exact mechanism is not clear. We selected ruthenium complexes [Ru(Phen)2(4idip)](ClO4)2 (Ru) to induce the generation of NO in cells. SeNPs–PEG–RGD@Ru (Se@RuNPs) were prepared by modifying selenium nanoparticles with PEG, RGD and Ru. Se@RuNPs can promote uptake by Human Umbilical Vein Endothelial Cells (HUVECs) and trace the internalization and biodistribution. Experiments showed that Se@RuNPs target the abundant neovascular network of inflammatory sites to induce NO and promote the apoptosis of HUVECs and inhibit the growth of new vessels in local tissue. Moreover, NO activates autophagy by modulating signaling pathways related to AMPKα and mTOR, increasing the flux of autophagy, inhibiting the activity of NF-κB-p65, and modulating the levels of inflammatory cytokines. The exact mechanism of the inflammatory response regulated by NO is revealed. Histopathological analysis showed that the Se@RuNPs effectively reduced synovitis, cartilage corrosion and inflammatory cytokine expression levels, achieving satisfactory therapeutic effects. These unexpected results provide an effective strategy for target treatment of RA.


 Please wait while we load your content...
Please wait while we load your content...