AMF responsive DOX-loaded magnetic microspheres: transmembrane drug release mechanism and multimodality postsurgical treatment of breast cancer†
Abstract
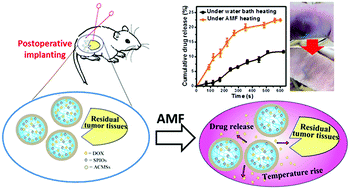
DOX-loaded magnetic alginate–chitosan microspheres (DM-ACMSs) were developed as a model system to evaluate alternating magnetic field (AMF)-responsive, chemo-thermal synergistic therapy for multimodality postsurgical treatment of breast cancer. This multimodality function can be achieved by the combination of DOX for chemotherapy, with superparamagnetic iron oxide nanoparticles (SPIONs) as magnetic hyperthermia agents and drug release trigger. Both moieties are encapsulated in ACMSs which also allow on-demand drug release. It is demonstrated that the optimized SPION content in DM-ACMSs is about 0.29 mg Fe, at which DM-ACMSs could exhibit the best hyperthermia performance. Under a remote AMF, DM-ACMs can quickly reach a 22.5% cumulative drug release in the tumor site within 10 min upon exposure under AMF, whereas only 0.2% DOX is released in the absence of AMF. Furthermore, a comparison study of AMF and water bath as heating source indicates that the cumulative drug release amount upon AMF exposure is twice that by water bath heating. Further analysis revealed that the AMF stimulated drug release is driven by both thermal and concentration gradient from inside to outside, which can be well-described by the coupling mechanism of mass and heat transfer using the Soret diffusion model. In vitro cytotoxicity tests on MCF-7 breast cancer cells show that the combined therapy based on DM-ACMSs leads to 95.5% cell death, about 1.5-fold and 1.1-fold higher than that of single magnetic hyperthermia or chemotherapy, respectively. The in vivo anti-tumor effect on tumor-bearing mice demonstrates that the residual tumor disappears in 12 days after chemo-thermal synergistic treatment using DM-ACMSs, and there is no recurrence in the entire experiment period (40 days) as compared to 25 days recurrence for single-modality treatment. Our results not only provide an innovative DM-ACMSs system as a stimuli-responsive, synergistic chemo-thermal therapy platform for efficient reduction in the recurrence of breast cancer, but also provide insight into the intricate interplay of the functional components in magnetic hydrogel microspheres.


 Please wait while we load your content...
Please wait while we load your content...