The effects of annealing barium niobium oxynitride in argon on photoelectrochemical water oxidation activity†
Abstract
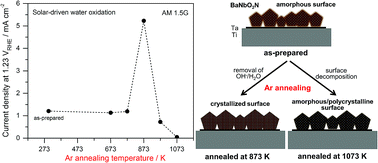
The n-type perovskite oxynitride BaNbO2N, which is capable of absorbing visible light up to a wavelength of 740 nm, was synthesized by thermal nitridation of Ba-rich crystalline Ba5Nb4O15. Mild nitridation of this starting oxide having a non-stoichiometric Ba/Nb ratio typically lowers the potential for reduction of Nb species but generates an amorphous surface. Herein, we report the effect of different annealing of BaNbO2N under inert Ar on its photoelectrochemical water oxidation activity. The surface properties of BaNbO2N, including its surface crystallinity and defect density, were found to be highly dependent on the annealing temperature, while the bulk crystallinity and optical properties were almost unaffected. At temperatures lower than 773 K, the annealing treatment had no effect on the inactive, amorphous nature of the as-prepared oxynitride. However, annealing at 873 K promoted surface crystallization of BaNbO2N by removing excess oxygen species generated during the synthesis process. The surface crystallization was completed by annealing for 1 h so that the photoanode using the annealed BaNbO2N exhibited a five times higher photocurrent density of over 5 mA cm−2 at 1.23 VRHE under simulated sunlight (AM 1.5G). Higher temperatures from 973 to 1073 K rather accelerated thermal decomposition of the oxynitride leading to release of nitrogen, accompanied by a gradual increase in surface defects and regeneration of an amorphous surface. The highly defective BaNbO2N photoanode produced almost negligible photocurrent density. As a result, the surface characteristics of BaNbO2N determine its water oxidation photoactivity, thus demonstrating that these surface properties must be carefully tuned for efficient solar-driven water oxidation.


 Please wait while we load your content...
Please wait while we load your content...