Selective atomic layer deposition of RuOx catalysts on shape-controlled Pd nanocrystals with significantly enhanced hydrogen evolution activity†
Abstract
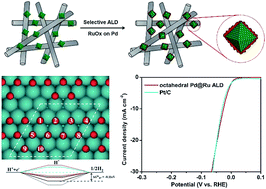
The electrochemical hydrogen evolution reaction (HER) is an emerging route for producing clean hydrogen. Recently, Ru-based catalysts have exhibited good activity towards the HER, making them attractive substitutes for Pt. However, the HER performance of Ru-based catalysts still cannot reach the same level as that of commercial Pt/C in acid solution. The atomic layer deposition (ALD) technique has proved to be an effective route for the preparation of noble metal catalysts on substrates. Herein, we successfully deposited RuOx catalysts on octahedral and cubic Pd seeds through an ALD process. Interestingly, we found that RuOx was selectively deposited on Pd particles when Pd/nitrogen-doped carbon nanotubes (NCNTs) were used as the substrate. Importantly, the as-prepared Pd@RuOx/NCNT showed comparable activity with state-of-the-art commercial Pt/C catalysts for the HER. The performance exceeds all of the current Ru-based nanocatalysts for the HER in acid solution. The X-ray absorption spectroscopy and X-ray photoemission spectroscopy analyses indicated that the RuOx shell consists of metallic Ru and Ru(IV). Density functional theory calculation results indicated that the Gibbs free energy of the intermediate state H* of octahedral Pd@RuOx was much lower than that of cubic Pd@RuOx, which resulted in the superior HER activity. This paper provides an example for the rational design of efficient catalysts through a combination of shape-controlled synthesis and the ALD technique.


 Please wait while we load your content...
Please wait while we load your content...