Heterointerface engineering of trilayer-shelled ultrathin MoS2/MoP/N-doped carbon hollow nanobubbles for efficient hydrogen evolution†
Abstract
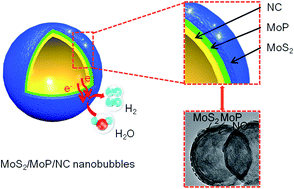
For efficient electrocatalysis, the rational construction of unique electrochemical interfaces is very important to enhance the intrinsic activity and expose more active sites. Herein, we demonstrate an atomic-migration-driven in situ thermal sulfurization–phosphorization strategy for the preparation of triple-layer-shelled hollow nanobubbles consisting of defect-rich ultrathin MoS2/MoP outer layers and a porous N-doped carbon inner layer (MoS2/MoP/NC) for efficient hydrogen evolution reaction (HER). In this method, (NH4)2MoS4/NaH2PO4-blended polymer nanospheres were prepared via aqueous-phase reaction, followed by a one-step thermal annealing process. During the thermal treatment, the MoS2 outer shell and NC inner layer were first formed at 500 °C; then the temperature was increased to 900 °C and the competitive reaction between the Mo atoms of the MoS2 species formed a strong driving force to transfer P species from the interior to the surface of the porous NC layer and form an intermediate layer of MoP. This strategy realized the formation of ultrathin MoS2/MoP/NC heterointerfaces with a high surface area (954.3 m2 g−1), abundant defect/edge sites, and improved electrocatalytic activity. In both acidic and alkaline solutions, the MoS2/MoP/NC hollow nanobubbles exhibited low overpotentials (151 and 208 mV) to drive a current density of 10 mA cm−2, small Tafel slopes (58 and 62 mV dec−1), and excellent stability for hydrogen production, respectively. This work provides a new route for the construction of active electrochemical heterointerfaces for efficient electrocatalysis.


 Please wait while we load your content...
Please wait while we load your content...