A Cu3P nanowire enabling high-efficiency, reliable, and energy-efficient low-voltage electroporation-inactivation of pathogens in water†
Abstract
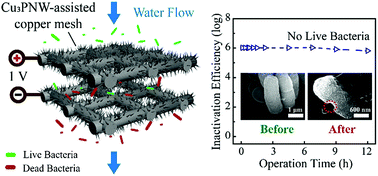
Pathogen infection has become the major reason for human morbidity and mortality in the world. However, common bacterial inactivation methods (e.g., chlorination, ultraviolet radiation, and ozonization) have significant drawbacks such as carcinogenic byproduct formation, energy intensiveness, and/or regrowth of pathogens. Nanowire-assisted low-voltage electroporation enables effective and energy-efficient bacterial inactivation. Here, we develop a new Cu3P nanowire-assisted copper mesh (Cu3PNW–Cu) electrode via an in situ growth followed by a phosphidation method and, for the first time, introduce the Cu3PNWs in the water purification process. An electroporation-disinfection cell (EDC) equipped with two such electrodes achieves superior bacterial inactivation performance (>6.0 log removal; no live bacteria in the effluent) with a low voltage of 1 V and a high flux of 2.0 m3 h−1 m−2. Under such operating conditions, the Cu3PNW–Cu electrode continuously treats water for 12 h while maintaining complete bacterial inactivation. The disinfection mechanism of electroporation guarantees an exceedingly low level of energy consumption: only 1.2 J for treating 1 L of water. To our knowledge, this is the lowest value obtained to date: >5 orders of magnitude lower than the typical energy consumption for bacterial inactivation using electroporation (150 kJ L−1).


 Please wait while we load your content...
Please wait while we load your content...