Elucidating the energy storage mechanism of ZnMn2O4 as promising anode for Li-ion batteries†
Abstract
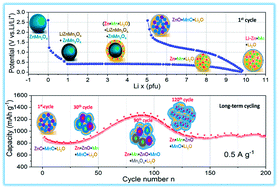
Tetragonal spinel ZnMn2O4 provides extremely high capacity as an anode for Li-ion batteries owing to a conversion-type mechanism. In this work, nanoparticle-composed layered ZnMn2O4 is synthesized using a co-precipitation method. Calcination parameters are optimized through thermal gravimetric analysis and in situ high temperature synchrotron X-ray powder diffraction. The ZnMn2O4 shows an initial lithiation capacity of ∼1400 mA h g−1 and a high reversible capacity of ∼900 mA h g−1 at a specific current of 0.5 A g−1. In situ synchrotron X-ray powder diffraction reveals phase evolution during the 1st cycle. An intermediate phase, tetragonal spinel LiZnMn2O4, is formed and coexists with the original ZnMn2O4 during the 1st lithiation. Electrochemical impedance spectroscopy applied at varying potentials during the 1st cycle provides evidence of the high Li+ diffusion coefficient and low resistance of the electrode in the lithiated state, which enables a high rate performance with 810 mA h g−1 at 1 A g−1 and 580 mA h g−1 at 2 A g−1. X-ray photoelectron spectroscopy reveals that the solid-electrolyte interphase is mainly composed of LiOH and Li2CO3, which can contribute additional capacity. In addition, an Mn(II)/Mn(III) redox reaction appearing during the 60th to 100th cycles is reported for the first time and could be another reason for the capacity increase upon cycling (the maximum capacity is ∼1250 mA h g−1 at the 90th cycle). This redox reaction is facilitated by the increase in the electronic conductivity upon cycling. Based on these investigations, fundamental insights into the energy storage mechanism of ZnMn2O4 conversion anodes in Li-ion batteries are clarified. This work can shed light on an understanding of other conversion-type electrode materials.


 Please wait while we load your content...
Please wait while we load your content...