Influence of dopant size and electron affinity on the electrical conductivity and thermoelectric properties of a series of conjugated polymers†
Abstract
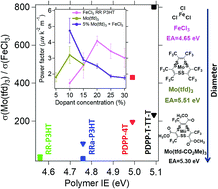
Chemical doping is widely used to manipulate the electrical and thermoelectric properties of organic semiconductors, yet intelligent design of polymer–dopant systems remains elusive. It is challenging to predict the electrical and thermoelectric properties of doped organic semiconductors due to the large number of variables impacting these properties, including film morphology, dopant and polymer energetics, dopant size, and degree of polaron delocalization. Herein, a series of dopants with varying sizes and electron affinities (EAs) are combined with polymers of differing ionization energies (IEs) to investigate how the difference between polymer IE and dopant EA influences the doping efficiency and electrical conductivity, and how the dopant size influences the thermoelectric properties. Our experiments demonstrate that at low doping levels the doping efficiency strongly depends on the difference between the polymer IE and dopant EA; the effectiveness of doping on increasing electrical conductivity drastically decreases at high loadings for the molybdenum dithiolene complexes, while FeCl3 remains effective at high loadings; and the large molybdenum complexes lead to more delocalized polarons as compared to FeCl3. To take advantage of the complementary doping characteristics of the molybdenum complexes and FeCl3, both dopants are employed simultaneously to reach high power factors at relatively low dopant concentrations.


 Please wait while we load your content...
Please wait while we load your content...