Electrodeposited single-crystalline PbCrO4 microrods for photoelectrochemical water oxidation: enhancement of minority carrier diffusion†
Abstract
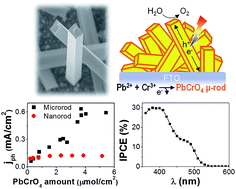
In this study, we report noticeable photoelectrochemical water oxidation on single-crystalline PbCrO4 microrods. Microrods are directly formed on fluorine-doped tin oxide via electrodeposition based on the electrochemical oxidation of Cr3+ to Cr6+ in the presence of Pb2+ in the electrolyte. The as-deposited PbCrO4 microrods are single-crystalline in the shape of a long rod with a 1 μm squared cross-section where a well-organized PbCrO4 growth is achieved with a low nucleation rate. The as-deposited PbCrO4 microrods absorb UV and visible light (λ < 540 nm), and utilize it for the photoelectrochemical reaction. The photoactivity of PbCrO4 microrods is greater than that of PbCrO4 nanorods, having originated from an enhanced minority carrier diffusion length increased by approximately 100 nm. PbCrO4 microrods with the help of Co–Pi decoration exhibit a noticeable photocurrent (0.5 mA cm−2 at 1.23 V vs. RHE under 1.5 AMG illumination) and high incident photon-to-current conversion efficiency (30% at λ = 400 nm) for photoelectrochemical water oxidation. Co–Pi decoration also effectively suppresses the formation of Pb4+ in PbCrO4, and improves the stability of the as-deposited PbCrO4 microrods with higher faradaic efficiency, as measured with scanning electrochemical microscopy. Furthermore, the control of electrodeposition potential is found to change the deposit composition between Pb and Cr, which leads to the transition from PbCrO4 to Pb2CrO5.


 Please wait while we load your content...
Please wait while we load your content...