Ionic activation via a hybrid IL–SSE interfacial layer for Li–O2 batteries with 99.5% coulombic efficiency†
Abstract
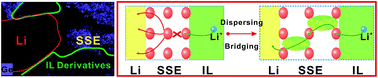
The open to the atmosphere nature of lithium–O2 batteries provides them with an inexhaustible supply of oxygen, resulting in them having a high specific energy, but this is a double-edged sword, as it also brings about the potential for explosion and drying out. Ionic liquids (IL) and solid-state electrolytes (SSE) are the most reliable systems in terms of avoiding these issues because they have near-zero vapor pressure and are generally non-flammable. However, no ILs or SSE lithium–O2 batteries have been operated under full discharge–charge conditions so far, that is, cycling the cells as a potential-controlled model. Herein, a lithium–O2 battery comprising an ether-functionalized ammonium IL electrolyte and carbon-free RuO2 cathode is reported, which has an average coulombic efficiency of 99.2% during repeated cycling. Furthermore, by incorporating a solid-state Li1.5Al0.5Ge1.5(PO4)3 (LAGP) film onto the Li metal anode, an in situ hybrid IL–SSE interfacial layer was formed, achieving 99.5% coulombic efficiency and significantly improving the battery cycleability over 100 cycles. The organic–inorganic hybrid layer exhibits ionic activation behavior through a dispersing redistribution and bridging process, revealing the beneficial interaction of IL and SSE on the cycling in safe IL–SSE-based Li–O2 batteries.


 Please wait while we load your content...
Please wait while we load your content...