A single ion conducting separator and dual mediator-based electrolyte for high-performance lithium–oxygen batteries with non-carbon cathodes†
Abstract
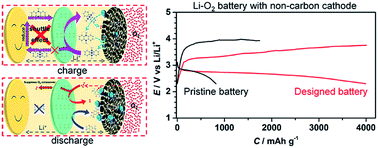
The application of non-carbon cathodes for mitigating side reactions in lithium–oxygen batteries faces the critical disadvantages of significantly reduced discharge capacity and unsatisfactory charge potential. Here, we break through these bottlenecks by introducing two synergistic redox mediators (RMs) into an electrolyte for lithium peroxide formation and reversible oxidation, and more importantly, by developing a single ion conducting Li+-Nafion separator to prohibit RM crossover towards the Li metal anode. The self-discharge and shuttle problems are avoided. The discharge capacity of the commercial ruthenium oxide-based non-carbon cathode can be greatly enhanced from 800 mA h g−1 to 3800 mA h g−1. The charge potential can be significantly reduced to 3.2 V, which is among the best levels reported. During the long-term cycling, the battery maintains a low charge overpotential of 0.24 V, indicating high stability. This first synergistic concept of designing RMs and a Li+-Nafion separator for advanced non-carbon lithium–oxygen batteries shows great potential for developing practical batteries with high capacity, energy efficiency and rechargeability.


 Please wait while we load your content...
Please wait while we load your content...