High efficiency electrochemical reduction of CO2 beyond the two-electron transfer pathway on grain boundary rich ultra-small SnO2 nanoparticles†
Abstract
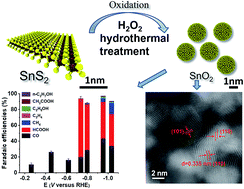
Well crystallized and interconnected SnO2 nanoparticles (<5 nm) were synthesized via oxidation of exfoliated SnS2 sheets. The SnO2 nanoparticles exhibit a high total faradaic efficiency (FE) of 97% towards electrochemical reduction of CO2 at −0.95 V vs. the reversible hydrogen electrode (RHE). The main product ratio of CO/HCOO− which intrinsically correlates to the surface SnOx/Sn ratio variation varies with the applied potential. Beyond CO and HCOO− products formed via the two-electron transfer pathway, hydrocarbons and oxygenates are produced. The formation of hydrocarbon (CH4) versus oxygenate (C2H5OH) depends on the choice of electrolyte (KOH vs. KHCO3), both of which can reach a maximal faradaic efficiency of 10%. The distinctive grain boundary and exposed corner/step sites in the interconnected SnO2 nanoparticles contribute to the high FE of CO2 reduction and unique selectivity.


 Please wait while we load your content...
Please wait while we load your content...