Tunable electronic coupling of cobalt sulfide/carbon composites for optimizing oxygen evolution reaction activity†
Abstract
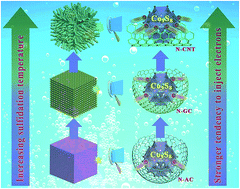
Hybrid nanocomposites consisting of non-precious transition-metal oxides/chalcogenides and a carbon matrix are of great interest for future applications in sustainable energy storage and conversion systems owing to their unique chemical and physical properties as well as the synergism. However, the construction of nanocomposites with tunable coupling effects to realize synergetic effects and optimal performance remains challenging. Herein, we develop a facile strategy for the synthesis of cobalt sulfide (Co9S8) nanoparticles encapsulated in the carbon matrix from a Prussian blue analogue (PBA) through a simultaneous sulfidation and carbonization process. With an increase in the degree of carbonization, the coordinated organic ligands (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) liberated from the PBA can be evolved as N-doped amorphous carbon (N-AC), N-doped graphitic carbon (N-GC) and N-doped carbon nanotubes (N-CNTs), respectively, generating the tunable electronic coupling between the Co9S8 and the carbon matrix. As an example, we show that the oxygen evolution reaction (OER) activity of such nanocomposites can be made comparable to the state-of-the-art catalytic properties of precious catalysts by optimizing the electronic coupling between the components so that electron injection from the N-doped carbon to the catalytically active site is greatly facilitated. Furthermore, the in situ formation of cobalt hydroxides on the surface of Co9S8 is also confirmed during the OER process, which might induce the interfacial effect, i.e. electron interplay, thus altering the OER catalytic activity. The current work provides new insights into the rational design of advanced hybrid nanocomposites for energy and environmental applications.
N) liberated from the PBA can be evolved as N-doped amorphous carbon (N-AC), N-doped graphitic carbon (N-GC) and N-doped carbon nanotubes (N-CNTs), respectively, generating the tunable electronic coupling between the Co9S8 and the carbon matrix. As an example, we show that the oxygen evolution reaction (OER) activity of such nanocomposites can be made comparable to the state-of-the-art catalytic properties of precious catalysts by optimizing the electronic coupling between the components so that electron injection from the N-doped carbon to the catalytically active site is greatly facilitated. Furthermore, the in situ formation of cobalt hydroxides on the surface of Co9S8 is also confirmed during the OER process, which might induce the interfacial effect, i.e. electron interplay, thus altering the OER catalytic activity. The current work provides new insights into the rational design of advanced hybrid nanocomposites for energy and environmental applications.


 Please wait while we load your content...
Please wait while we load your content...