Phosphorene oxides as a promising cathode material for sealed non-aqueous Li–oxygen batteries†
Abstract
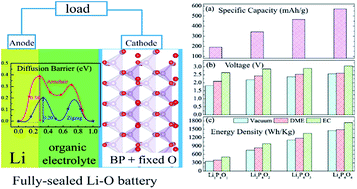
A new type of two-dimensional (2D) material, phosphorene oxides (POs) in four surface oxidation states (P4O1, P4O2, P4O3 and P4O4), is evaluated as a potential cathode for sealed Li–oxygen batteries by combining the first-principles method with molecular dynamics simulations and implicit solvent models. The system can be sealed because, unlike open Li–air batteries, there is no need to take oxygen from air. Our simulations reveal that: (1) by forming a maximum number of Li–O bonds, Li can be tightly chemisorbed on the surface of POs with a large binding energy ranging from −2.32 to −3.72 eV and a charge transfer of ∼0.9e from Li to O. (2) The diffusion of Li on POs is strongly anisotropic as the barrier along the armchair direction is nearly two times higher than that along the zigzag direction. The barrier dramatically decreases to 0.2 eV with increasing oxidation, indicating a high diffusivity. (3) For the P4O4 cathode, the thermodynamically stable, fully discharged product has a stoichiometry of Li : P : O = 1 : 1 : 1, achieving a specific capacity of 570.56 mA h g−1, an average open-circuit voltage of 2.55 V and an energy density of 1457 W h kg−1. The energy density can be further increased by considering solvent effects. More polar cyclic ethylene carbonate (EC) solvent brings about a larger energy density enhancement than less polar linear 1,2-dimethoxyethane (DME). (4) Semiconductor-to-metal transition occurs within POs after Li adsorption, facilitating the electrical transport in PO-based batteries. A combination of strong Li adsorption, high diffusivity, and good conductivity makes POs great candidates for future fully sealed Li–oxygen batteries.


 Please wait while we load your content...
Please wait while we load your content...