Promoting sulfur adsorption using surface Cu sites in metal–organic frameworks for lithium sulfur batteries†
Abstract
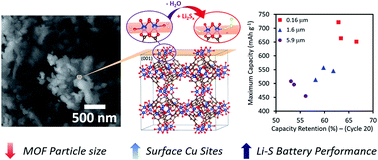
Despite the promise of high energy density in lithium sulfur (Li–S) batteries, this technology suffers from poor long-term stability due to the dissolution of polysulfides upon battery cycling. Metal–organic frameworks (MOFs) are shown to be effective cathode materials for Li–S batteries, but the nature of sulfur–host interactions in these porous materials is not well-understood. Herein, we provide a detailed analysis on material and chemical properties that have direct influence on sulfur adsorption and battery performance. Favorable binding sites in CuBTC, a MOF proven promising for sulfur adsorption, are identified and characterized by a combination of theoretical, thermogravimetric, spectroscopic, and electrochemical experiments. By manipulating MOF particle size, we further demonstrate that a high density of Cu-rich surface defects drastically improves both maximum discharge capacity and polysulfide retention. Battery cycling data illustrates the significance of these surface Cu sites for the uptake of dissolved polysulfides, which mitigates irreversible capacity loss. In the wider scope of materials development, our findings suggest the use of carefully engineered surface defects in inorganic nanomaterials may enhance sulfur capture in Li–S batteries. This study thus advances chemical understanding towards rational design of porous materials with great implications for energy storage, sulfur removal, chemical sensing, and environmental remediation.


 Please wait while we load your content...
Please wait while we load your content...