Complex hydrides as thermal energy storage materials: characterisation and thermal decomposition of Na2Mg2NiH6†
Abstract
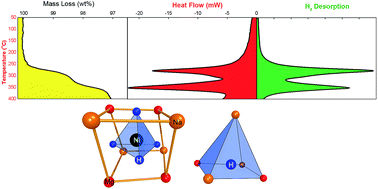
Complex transition metal hydrides have been identified as being materials for multi-functional applications holding potential as thermal energy storage materials, hydrogen storage materials and optical sensors. Na2Mg2NiH6 (2Na+·2Mg2+·2H−·[NiH4]4−) is one such material. In this study, the decomposition pathway and thermodynamics have been explored for the first time, revealing that at 225 °C, hydrogen desorption commences with two major decomposition steps, with maximum H2 desorption rates at 278 and 350 °C as measured by differential scanning calorimetry. The first step of decomposition results in the formation of Mg2NiHx (x < 0.3) and NaH, before these compounds decompose into Mg2Ni and Na, respectively. PCI analysis of Na2Mg2NiH6 has determined the thermodynamics of decomposition for the first step to have a ΔHdes and ΔSdes of 83 kJ mol−1 H2 and 140 J K−1 mol−1 H2, respectively. Hydrogen cycling of the first step has been achieved for 10 cycles without any significant reduction in hydrogen capacity, with complete hydrogen desorption within 20 min at 395 °C. Despite the relatively high cost of Ni, the ability to effectively store hydrogen reversibly at operational temperatures of 318–568 °C should allow this material to be considered as a thermal energy storage material.


 Please wait while we load your content...
Please wait while we load your content...