Understanding the crucial role of local crystal order in the electrocatalytic activity of crystalline manganese oxide†
Abstract
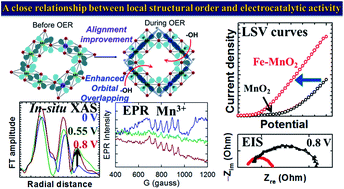
The structure–property relationship in transition metal oxides is of crucial importance in designing and synthesizing economically feasible high-performance electrocatalysts. Since cation substitution allows to finely tailor the atomic arrangement, structural distortion, and electrocatalytic performance of transition metal oxides, a relationship between local structural order and electrocatalytic activity in crystalline manganese oxide can be systematically investigated by in situ X-ray absorption, electron paramagnetic resonance, and electrochemical impedance spectroscopic analyses for unsubstituted and Fe-substituted α-Mn1−xFexO2 during the oxygen evolution reaction (OER). The substitution of Mn with Fe is quite effective in improving the OER activity of α-MnO2 to reach a small overpotential of 0.40 V at 10 mA cm−2. Under OER conditions, the Fe substitution improves the local structural order of MnO6 octahedra in the α-MnO2 lattice, thus leading to a significant enhancement of charge transport kinetics. Since the Fe substitution induces only a limited alteration of the electronic structure and the substituted Fe ion itself shows only a negligible contribution to the OER activity, the excellent OER functionality of Fe-substituted α-Mn1−xFexO2 is attributable mainly to the improvement of local structural ordering upon Fe substitution. The present study underscores the crucial role of local structural order in optimizing the electrocatalytic functionality of crystalline transition metal oxides.


 Please wait while we load your content...
Please wait while we load your content...