Surface stoichiometry manipulation enhances solar hydrogen evolution of CdSe quantum dots†
Abstract
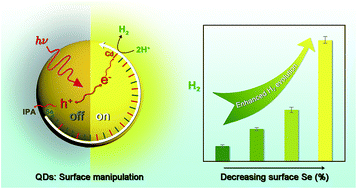
Surface stoichiometry is a sensitive parameter affecting the decay dynamics of photogenerated hole–electron pairs of QDs. However, the effect of this manipulation on artificial photocatalytic H2 evolution is unclear. Here, we report that surface stoichiometry manipulation is a facile and feasible approach for enhancing H2 photogeneration of QDs. In the absence of an external cocatalyst, a decrease in the surface Se ratio of CdSe QDs from ∼16.7% to ∼4.9% gives a more than 10-fold increase in solar H2 evolution. Taking Ni(II) as an external cocatalyst, CdSe QDs with a surface Se ratio of ∼4.9% can produce ∼1600 ± 151 μmol H2 gas during 27 h of visible-light irradiation, giving a total turnover number of (1.24 ± 0.12) × 105 on CdSe QDs and an apparent quantum yield of 10.1%, which is about 8 times that of CdSe QDs with a surface Se ratio of ∼16.7% under the same conditions. Mechanistic insights obtained by a combination of steady-state and time-resolved spectroscopic techniques indicate that surface stoichiometry exerts a significant influence on the exciton kinetics of CdSe QDs: a higher ratio of surface Se would increase the possibility of exciton recombination through hole trapping, thus depressing the performance of solar H2 evolution.


 Please wait while we load your content...
Please wait while we load your content...