Electrodeposited amorphous Co–P–B ternary catalyst for hydrogen evolution reaction†
Abstract
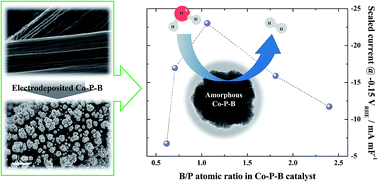
The catalytic activity of amorphous Co–P–B catalysts electrodeposited on carbon paper (CP) for the hydrogen evolution reaction (HER) was investigated in aqueous 0.5 M H2SO4 electrolyte. Based on the measured double layer capacitance (Cdl) and HER activity, it was found that the HER intrinsic activity exhibited volcano behavior when plotted as a function of the B/P ratio in the Co–P–B catalyst. It was revealed that based on the electronic structures of the elements, electron transfer among the three elements is a significant factor for enhancing the intrinsic activity for the HER. The highest activity was obtained with a B/P ratio of ∼1, where electron transfer among the three elements was maximized.


 Please wait while we load your content...
Please wait while we load your content...