Enhanced visible light photocatalytic activity of TiO2 assisted by organic semiconductors: a structure optimization strategy of conjugated polymers†
Abstract
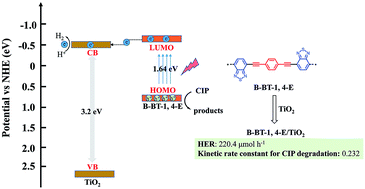
The search for high-efficient TiO2-based heterojunction photocatalyst for photocatalytic H2 evolution and pollutant removal remains a great challenge. In this study, linear conjugated polymer B-BT-1,4-E consisting of alternating electron donor and electron acceptor was incorporated on the surface of TiO2 to form a binary composite in the facile in situ strategy. B-BT-1,4-E/TiO2 exhibited superior photocatalytic activity under visible light irradiation (λ ≥ 420 nm). The hydrogen evolution rate (HER) of 16.7% B-BT-1,4-E/TiO2 reached 220.4 μmol h−1 without additional noble metal (Pt), and the optimized 13.3% B-BT-1,4-E/TiO2 also displayed outstanding CIP degradation efficiency with a kinetic constant of 0.232. Based on extensive characterization results from UV-Vis diffused reflectance spectra (DRS), electron spin resonance (ESR), photocurrent, electrochemical impedance spectroscopy (EIS) and photoluminescence (PL), the enhanced photocatalytic activity of B-BT-1,4-E/TiO2 heterojunction can be attributed to broader visible light absorption range, faster charge separation and transfer. Moreover, a reasonable photocatalytic mechanism is also proposed. In comparison to reported CMP(BBT)/TiO2, HER and kinetic constants of CIP degradation with B-BT-1,4-E/TiO2 as the photocatalyst were increased by 7.3 times and 3.1 times, respectively. This study demonstrated that intrinsic merits of conjugated polymers made them promising candidates for exploring more efficient organic semiconductor–inorganic semiconductor heterojunction photocatalysts.


 Please wait while we load your content...
Please wait while we load your content...