TiO2–P3HT:PCBM photoelectrochemical tandem cells for solar-driven overall water splitting†
Abstract
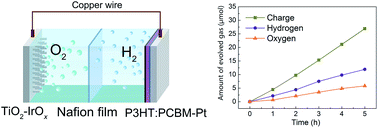
The existing photoelectrodes for photoelectrochemical (PEC) cells are almost based on oxide semiconductors. Compared with n-type oxide photoanodes, p-type oxide photocathodes are rather rare and most are not suitable for unassisted water reduction because of their inappropriate band edge positions. Thus, it is highly desirable to exploit more kinds of p-type semiconductors for Z-scheme PEC cells. Commonly, organic semiconductors are p-type with a more negative band edge, which will give more options for photocathodes and provide new paths for sustainable overall water splitting. Herein, we report a PEC cell in which TiO2 and the poly(3-hexylthiophene):[6,6]-phenyl-C61-butyric acid methyl ester bulk heterojunction (P3HT:PCBM BHJ) are connected by copper wires to achieve solar-driven overall water splitting in the absence of an external bias. The photocurrent density of the TiO2–P3HT:PCBM system is about 145 μA cm−2 and remains constant in a 9 h test duration under continuous illumination. The average H2 and O2 evolution rates of this system are about 2.38 μmol h−1 and 1.16 μmol h−1, respectively. The charge-to-chemical faradaic efficiency is 88.7%. These results confirm that a PEC cell for overall water splitting can be achieved by coupling the most representative inorganic semiconductor TiO2 and the most considered organic material P3HT:PCBM.


 Please wait while we load your content...
Please wait while we load your content...