Carbon-supported metal nanodendrites as efficient, stable catalysts for the oxygen reduction reaction†
Abstract
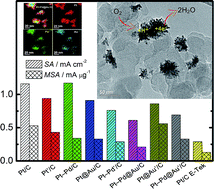
The search for efficient, stable electrocatalysts for the oxygen reduction reaction (ORR) has received increased attention, given the need to speed up this reaction in fuel cells. This article reports the one-pot synthesis of novel metal nanodendrites (MNDs) of Pt or Pt–Pd alloy surface-covering patterns that, supported on Vulcan Carbon XC-72, effectively catalyzed the ORR. The surface of Vulcan Carbon XC-72 exhibits raised plains interspersed with ribbed troughs, in a pattern energetically favorable to metal precipitation (deposition) into the ribbed troughs. This produces MND/C structures that are strongly catalytic toward the ORR. Mass-specific activity (MSA) of 0.56 mA μg−1 and specific activity (SA) in the 1.17–1.35 mA cm−2 range are noteworthy findings for Pt/C, Pt@Au′/C, and Pt–Pd/C MND electrocatalysts at 0.9 ViR-free, using platinum-group metal (PGM) loadings as low as 26 μg cm−2—better values, therefore, than the United States Department of Energy (DOE) targets for MSA (0.44 A mgPt−1) and SA (0.72 mA cm−2 at 0.9 ViR-free) for electrocatalysts used in portable applications to be marketed in 2017, and for cathode areal PGM loadings (<50 μgPGM cm−2), as well as better than the commercial E-Tek Pt/C (20% Pt mass) catalyst.
- This article is part of the themed collection: Materials and Nano Research in Brazil


 Please wait while we load your content...
Please wait while we load your content...