From anti-perovskite to double anti-perovskite: tuning lattice chemistry to achieve super-fast Li+ transport in cubic solid lithium halogen–chalcogenides†
Abstract
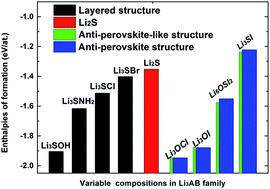
Using a materials genome approach on the basis of density functional theory, we have formulated a new class of inorganic electrolytes for the fast diffusion of Li+ ions by fine-tuning the lattice chemistry of anti-perovskite structures. Systematic modelling was carried out to determine the structural stability and ion transport characteristics in Li3AX-based cubic anti-perovskites by alloying on the chalcogen lattice site (A) and alternative occupancy of the halogen site (X). In addition to identifying effective ways of reducing diffusion barriers for Li+ ions in anti-perovskite phases via the suitable designation of lattice occupancy, the current theoretical study has led to the discovery and synthesis of a new phase with a double-anti-perovskite structure, Li6OSI2 (or Li3O0.5S0.5I). This new compound has a fairly low activation barrier for Li+ diffusion, together with a wide energy band gap to hinder the conduction of electrons.


 Please wait while we load your content...
Please wait while we load your content...