Fe/Fe3C@C nanoparticles encapsulated in N-doped graphene–CNTs framework as an efficient bifunctional oxygen electrocatalyst for robust rechargeable Zn–air batteries†
Abstract
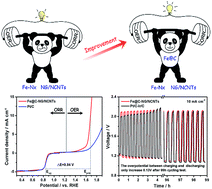
3d transition metals or their derivatives encapsulated in nitrogen-doped nanocarbon show promising potential in non-precious metal oxygen electrocatalysts. Herein, we describe the simple construction of a bifunctional oxygen electrocatalyst with a “framework-active sites” structure, namely Fe/Fe3C@C (Fe@C) nanoparticles encapsulated in 3D N-doped graphene and bamboo-like CNTs (Fe@C–NG/NCNTs). The Fe@C structure provides additional electrons on the carbon surface, promoting the oxygen reduction reaction (ORR) on adjacent Fe–Nx active sites. The 3D NG hybrid with a bamboo-like CNTs framework facilitates fast reactant diffusion and rapid electron transfer. The optimized sample displays excellent ORR and oxygen evolution reaction (OER) activity, with a potential difference of only 0.84 V; this places it among the best bifunctional ORR/OER electrocatalysts. Most importantly, Zn–air batteries using Fe@C–NG/NCNTs as the cathode catalyst deliver a peak power density of 101.2 mW cm−2 and a specific capacity of 682.6 mA h g−1 (energy density of 764.5 W h kg−1). After 297 continuous cycle tests (99 h), the rechargeable batteries using Fe@C–NG/NCNTs show a voltage gap increase of only 0.13 V, almost half that of Pt/C + Ir/C (0.22 V) under the same conditions. This work provides new insight into advanced electrocatalysts utilizing the structural features of host nanocarbon materials and guest active species toward energy conversion.
- This article is part of the themed collections: 10th Anniversary: Most popular articles and International Year of the Periodic Table: Elements for Next Generation Batteries


 Please wait while we load your content...
Please wait while we load your content...