Oxygen transport phenomena in (La,Sr)2(Ni,Fe)O4 materials†
Abstract
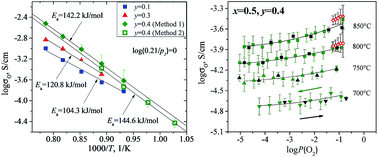
Oxygen-ion transport in La2−xSrxNi1−yFeyO4+δ complex oxides (x = 0.5, y = 0.1–0.4 and x = 0.8, y = 0.1, 0.2, and 0.4) was studied by steady-state oxygen permeation and modified Hebb–Wagner polarization methods. The samples were obtained by a decomposition of citrate–nitrate precursors followed by three times subsequent annealing at 1100 °C in air, pelletizing and sintering at 1300–1350 °C for 20 h in air. The phase purity of the samples was confirmed by an X-ray diffraction (XRD) method. Energy dispersive X-ray (EDX) analysis of the pellets after the steady-state oxygen permeation measurements revealed negligible diffusion of dopant cations and an almost constant oxygen gradient along the samples' thickness. Strontium was shown to decrease the oxygen permeation through the overstoichiometric La2−xSrxNi1−yFeyO4+δ (δ > 0) membranes and to increase it for the oxygen deficient ones (δ < 0). Iron promoted bulk diffusion in all studied samples. The oxygen surface exchange in the series La1.5Sr0.5Ni1−yFeyO4+δ was slightly deteriorated by iron doping up to y = 0.3 but significantly enhanced at y = 0.4. The values of oxygen-ion conductivity calculated from the oxygen permeation data were in good agreement with those measured by the modified Hebb–Wagner polarization method. The highest oxygen permeation flux was obtained for the La1.5Sr0.5Ni0.6Fe0.4O4+δ membrane. La1.5Sr0.5Ni0.6Fe0.4O4+δ showed lower oxygen-ion conductivity compared with undoped La2NiO4+δ, but substantially improved oxygen surface exchange. The latter factor plays a crucial role in the cathode performance of the studied La2NiO4+δ-based oxides and makes La1.5Sr0.5Ni0.6Fe0.4O4+δ a promising candidate for IT-SOFC application.
- This article is part of the themed collection: Advances in Solid State Chemistry and its Applications


 Please wait while we load your content...
Please wait while we load your content...